Stargardt Disease Treatment with Topical Acetylcholinesterase Inhibitor Medication, Low Dose Echothiophate Iodide, Patient 61
Gerard M Nolan, MD September 2021
Abstract
Purpose
To demonstrate visual acuity and color vision improvement realized by a Stargardt disease patient resulting from topical cholinergic medication (TCM).
Observations
Early and sustained improvement in both visual acuity and color vision in a patient with Stargardt disease. The improvement was a result of topical application of low dose echothiophate iodide (0.015%). The gains in visual acuity and color vision have been maintained through a period of observation of one year.
Conclusions and Importance
TCM, low dose echothiophate iodide therapy produced significant and sustained gains in BCVA and color vision for this patient. This compares favorably to the natural degradation to legal blindness in untreated Stargardt disease patients. Low dose echothiophate iodide therapy potentially provides a treatment for Stargardt disease where none was previously available.
Introduction
Today, Stargardt disease is the most common inherited single-gene retinal disease. It usually has an autosomal recessive inheritance caused by mutations in the ABCA4 gene.1 Stargardt disease inevitably leads to significant and irreversible loss of vision. Cholinergic stimulation (the parasympathetic nervous system) uses acetylcholine (ACh) at its receptors and synapses and is regulated by the enzyme acetyl cholinesterase. Echothiophate iodide inhibits this enzyme acetyl cholinesterase and allows for the increase of endogenous acetylcholine.
This case history presents evidence of early and sustained improvement in visual acuity and color vision in a Stargardt Disease patient. The improvement was a result of the application of TCM, low dose echothiophate iodide (0.015%).
Case Report / Findings
The present case involves a female patient diagnosed with Stargardt disease at age 19 in 1994. This individual presented with no familial history of blindness or retinal disease. The patient wore glasses from age 6 and contact lenses as a teenager with moderate myopia (-6.00 diopter). The past medical history was significant for allergy to penicillin and seasonal allergies. The patient’s identical twin (Patient 247) was diagnosed with Stargardt disease 6 months before her diagnosis. At the time of her twin sister’s diagnosis the patient had no symptoms except for a tiny dot appearing in the middle of each word while reading. On the insistence of her sister’s retinal vitreal specialist, she was examined. At the consultation, similar retinal pathology was diagnosed. She was told she would go blind and that nothing could be done about it. The patient resumed her university studies. At the beginning of the semester, she sat in the last row of the classroom, by the end of the semester she could not see the blackboard from the first row of the classroom.
Visual acuity continued a steady progressive decline until age 23, at which point the patient experienced an abrupt drop in both central vision and night vision and ceased driving. In 1997, the patient and her sister presented at Baylor University Medical Center for genetic testing. Test results revealed the presence of the ABCA4 gene consistent with Stargardt disease in both patients.
The patient initially presented for baseline testing at age 31 on March 7, 2006. The patient is included in a 20 year, 309 Stargardt disease patient study. The patient is also one of 22 patients in a one year, controlled proof-of-concept (POC) trial for Stargardt disease under the oversight of an Institutional Review Board (IRB).2
This ophthalmic clinical research was designed by PharmaLogic Development, Inc.3
Treatment Protocol
Prior diagnosis of Stargardt disease was verified via fluorescein-based angiogram. Best corrected visual acuity (BCVA) was established using the EDTRS system for distance and near vision and converted to logMAR for analysis. Color vision was documented by the Ishihara 10 plate test. The fluorescein-based angiogram revealed RPE dropout in both temporal and nasal retina. The patient was classified as RPE Atrophy Stage IV. The World Health Organization (WHO) visual impairment category was severe in the right eye and moderate in the left eye for distance vision and moderate in both eyes for near vision.
Following the initial consultation (visit 1), the patient returned the next day (visit 2.1) for retesting of baseline BCVA. The patient then received one drop of dilute (0.015%) echothiophate iodide in the lower cul-de-sac of both eyes and instructed to sit with eyes closed for one hour (visit 2.2). The patient spontaneously reported an increase in overall vision brightness. BCVA improved as follows,
Distance right eye improvement | 8 letters (1.6 lines) |
Distance left eye improvement | 3 letters (0.6 lines) |
Near right eye improvement | 8 letters (1.6 lines) |
Near left eye improvement | 6 letters (1.2 lines) |
Color vision remained unchanged.
The patient was instructed in the proper self-administration of eye drops. The dosage used for treatment of this patient was one drop of 0.015% echothiophate iodide, administered to each eye every two days, immediately prior to sleep thus minimizing runoff due to eye movement and blinking. The source for the eye drops was Wyeth-Ayerst, Philadelphia, PA. The product, Phospholine Iodide®, included both echothiophate iodide and its buffered diluent. The patient returned every month for the first three months and every three months for the next nine months.
Results
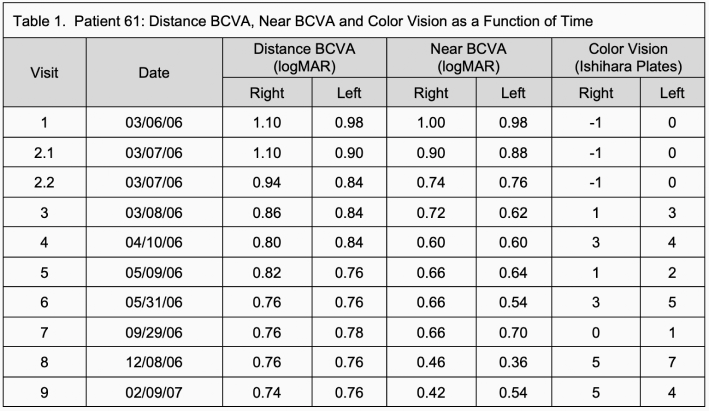
The pre-treatment examination (visit 1) determined that the patient's right eye distance BCVA was 20/250 (1.10 logMAR) and left eye was 20/200+1 (0.98 logMAR), eccentric fixation. Right eye near BCVA was 20/200 (1.00 logMAR). and the left eye 20/200+1 (0.98 logMAR). The patient’s gaze was eccentric with particular difficulty in near vision focus. Color vision was contrast-only, -1 plate (missed control plate) in the right eye and zero plates in the left eye.
Examination 24 hours after initial treatment (visit 3) revealed mild pupil narrowing of 1 mm (4 to 3 mm). The patient's right eye distance BCVA had improved to 20/160+2 (0.86 logMAR). Left eye distance BCVA improved to 20/125-2 (0.84 logMAR). Near BCVA in the right eye improved to 20/100-2 (0.74 logMAR) and to 20/125-2(0.76 logMAR) in the left. Color vision in right eye improved to 1 plate plus control plate and to 3 plates in the left.
The patient tolerated the medicine without any side effects. None of the complaints of ocular burning, redness, irritation, dimming or blurring of vision, lacrimation or brow-ache associated with the traditional 0.25% and 0.125% concentrations of echothiophate iodide treatment of glaucoma have been reported by this patient.
Within the first month of therapy (visit 4), distance vision BCVA improved to 20/125 (0.80 logMAR) in the right eye and 20/125-2 (0.84 logMAR) in the left eye. Near BCVA improved to 20/80 (0.60 logMAR) in both eyes. Color vision improved to 3 plates in the right eye and 4 plates in the left eye.
At one year (visit 9) distance BCVA was 20/100-1 (0.74 logMAR) in the right eye and 20/125+2 (0.76 logMAR) in the left eye. Near BCVA improved to 20/50-2 (0.42 logMAR) in the right eye and 20/63-2 (0.54 logMAR) in the left. Color vision was 5 plates in the right eye and 4 plates in the left eye.
Table 1 summarizes the patient’s distance and near visual acuity and color vision for the duration of the study.
Discussion
Dilute echothiophate iodide provided an effective, non-surgical therapy for this Stargardt disease patient. After visit 3, the patient left the office and could identify a green, John Deere tractor parked across the street. While having lunch at a local mall, she remarked that she could see the other diners, barstools, bottles of liquor and glasses full of ice cubes reflecting under the lights. Arriving home, she was able to discern lights and lines on the runway.
Returning home, she noticed that her house had popcorn ceilings and the fabrics of her furniture and curtains were outdated. She spent the next several months redecorating her home. She noticed that from her windows she was able to see into the neighbor’s apartment windows. She said she no longer felt comfortable parading around the room wearing only a towel after bathing. She remarked that she was able to see the curls in her children’s hair and their long eyelashes. She was able resume driving and reclaimed her independence. She summarized her treatment success by stating “The treatment is like turning on windshield wipers on a rainy day, everything becomes clearer and vibrant, ever since [starting treatment] I have been back to a normal functioning life.”
The patient’s vision gains have restored her independence and allowed her to be a more active parent. She has become an advocate for individuals suffering from blindness due to macular degeneration and Stargardt disease. For the last 15 years, the patient has been followed by her local vitreal retinal specialist. The patient has maintained the vision improvement observed during the treatment period. The patient continues to be independent, an active parent and maintains a current driver’s license.
In summary, this case study presents evidence of BCVA and color vision improvement for this Stargardt disease patient resulting from the application of low dose echothiophate. The treatment resulted in a marked improvement of quality-of-life.
Conclusions
Stargardt disease is one of a number of degenerative retinal diseases, including dry age-related macular degeneration and retinitis pigmentosa, which may respond to TCM therapy. If this turns out to be the case, this therapy may hold the potential to lessen the disability from a number of blinding retinal diseases.
The exact mechanism of action of echothiophate iodide in Stargardt disease patients is not yet known. Perhaps, dilute echothiophate iodide makes endogenous ACh more available to diseased neuroreceptor populations across the retina. Increased ACh levels may amplify the synaptic potential of the surviving photoreceptors and ganglion cells, making it possible for these reduced populations to achieve threshold and resume the propagation of visual information to the brain. This effect would parallel previously described mechanisms of drug action in other neurological diseases, such as Parkinsonism, Alzheimer’s Disease and Clinical Depression.
Topical cholinergic medications, echothiophate iodide, pilocarpine, etc., have been a staple to treat glaucoma and strabismus. Recently, the use of several different cholinergic therapeutics has been expanded to include treatment of presbyopia. This case study illustrates a single Stargardt disease patient treated with a cholinergic medication, i.e., low dose (0.015%) echothiophate iodide. The patient showed initial and long term improvement which has been maintained through a period of observation of more than 15 years. The treatment resulted in a marked improvement of the patient’s quality-of-life.
This compares favorably to the natural degradation of visual acuity often leading to legal blindness caused by this untreatable disease. Hopefully, the ophthalmology community will become aware of this Stargardt disease treatment regimen and add it to their tools for the treatment of this disease.
Patient consent
Written informed consent was obtained from the patient for publication of this case report. This report does not contain any personal identifying information.
Notes
- Michaelids M, Hunt DM, Moore AT. The genetics of inherited macular dystrophies. J Med Genet. 2003;40(9):641-650.
- IRB file number - 05175-01 (November 23, 2005) A Pilot, open-label study of low dose ocular echothiophate iodide for the treatment of Stargardt’s [sic] Disease. Principal Investigator Number - 5575-001. Sponsor - Independent Review Consulting, Inc. (IRC), Corte Medera, California
- PharmaLogic Development, Inc, San Rafael, California Ophthalmic Research and Development
Abbreviations and Acronyms:
ETDRS = Early Treatment Diabetic Retinopathy Study; IRB = International Review Board; RPE = retinal pigment epithelium; TCM = topical cholinergic medication; WHO = World Health Organization
 Return home
Return home